the Creative Commons Attribution 4.0 License.
the Creative Commons Attribution 4.0 License.
Vulnerability of carbon in subalpine soils in the face of warmer temperatures
Dario Püntener
Philipp Zürcher
Tatjana C. Speckert
Carrie L. Thomas
Guido L. B. Wiesenberg
Alpine and subalpine soils are significant reservoirs of labile carbon (C) and are highly sensitive to warming, yet the mechanistic interactions between temperature and organic inputs are poorly understood. A one-year laboratory incubation was conducted with mineral surface soils from a subalpine pasture and an adjacent coniferous forest site. Soil samples were incubated in closed jars at three different temperatures: current growing season temperature (12.5 °C), and two increased temperature treatments (16.5 and 20.5 °C). To assess decomposition differences between litter and native soil organic matter (SOM), 13C-labelled plant litter was added to a subset of the jars. CO2 production, δ13C partitioning, and phospholipid fatty acid (PLFA) profiles were used to quantify soil organic matter (SOM) and litter decomposition, and to assess microbial dynamics. Warming increased total CO2 respiration by 15 %–37 % in pasture and 12 %–33 % in forest soils, with strongest stimulation in litter-amended soils. Positive priming of native soil organic matter (SOM) peaked within one week (up to +77 % over controls) and declined to near zero after one month. Cumulative litter-induced respiration (LIR) was highest at 16.5 °C (+6 %–10 % vs. 12.5 °C) in both soils, coinciding with maximum microbial biomass; 20.5 °C reduced microbial biomass by up to 25 % and accelerated 13C label loss. The response of pasture soils was more rapid and pronounced compared to forest soils, which exhibited slower, more sustained responses. PLFA profiles revealed warming-induced declines in Gram+ and Gram− bacteria and increased cyclopropyl markers at high temperature. These findings show that even moderate warming can accelerate C loss from subalpine soils, with vegetation history and microbial traits modulating both rate and timing of decomposition.
- Article
(1012 KB) - Full-text XML
-
Supplement
(503 KB) - BibTeX
- EndNote
Soil organic matter (SOM) is a major component of the global carbon (C) cycle, containing more C than the atmosphere and terrestrial plant C pool combined (Schmidt et al., 2011; Friedlingstein et al., 2025). Due to generally cold temperatures and short growing seasons, soils in alpine regions tend to accumulate thick organic layers and store high stocks of C (Budge et al., 2011; Bonfanti et al., 2025). These low temperatures slow down microbial decomposition, causing SOM to accumulate in a labile, easily decomposable form (Djukic et al., 2010). As a result, alpine and subalpine SOM is particularly sensitive to warming (Chersich et al., 2015). A modest rise in soil temperature could greatly enhance microbial activity and thus CO2 release from SOM, potentially creating a positive climate feedback (García-Palacios et al., 2021; Soong et al., 2021; Chen et al., 2024). Currently, mountain regions are warming faster than lowlands (Rogora et al., 2018; Hock et al., 2019). At the same time, snow cover is declining (Klein et al., 2016), the growing season is prolonged (Rogora et al., 2018) and vegetation is shifting because treelines are moving upward and pastures are being abandoned (Gehrig‐Fasel et al., 2007; Hagedorn et al., 2019). As a result, the fate of SOM in alpine soils under ongoing climate change is a critical open question.
Large areas in alpine and subalpine regions are covered by grasslands (Zehnder et al., 2020), either naturally due to cold temperatures at high elevations above the treeline or due to human management (Schwörer et al., 2015). Recent changes leading to shifts in vegetation are driven by warming temperatures as well as the abandonment of mountain pastures (Gehrig‐Fasel et al., 2007). The most evident change is shrub encroachment and afforestation of former pasture landscapes. Similar changes are also occurring in natural grasslands near the rising treeline (Hagedorn et al., 2019; Zehnder et al., 2020). These vegetation shifts in alpine regions alter quality and quantity of C inputs into soils and SOC stocks (Gehrig‐Fasel et al., 2007; Hagedorn et al., 2019). The up-slope movement of shrubs and trees introduces more woody tissues and generally thicker organic layers, altering SOM composition (Speckert et al., 2023). This also strongly influences soil microbial communities, as often significant shifts in bacterial and fungal populations can be observed following afforestation (Gunina et al., 2017b). Resulting changes are variable, and the implications for SOM stability and the sensitivity to warming are still unclear. Differences in microbial community composition between vegetation types can affect decomposition speed, with pasture soils often hosting more copiotrophic microorganisms adapted to fast cycling of labile C, and forest soils containing more microorganisms specialized in degrading complex organic matter (Solly et al., 2014; Kramer and Gleixner, 2008; Zhang et al., 2016; Lu et al., 2017). These contrasts are consistent with stronger warming-induced SOC losses reported in forests relative to grasslands (Poeplau et al., 2020). We therefore compare a subalpine pasture soil and a forest soil from the same landscape setting to assess whether contrasts consistent with vegetation history can be detected in soil organic matter dynamics and carbon input quality.
Mechanistically, such vegetation shifts are expected to affect SOM dynamics through a linked sequence of changes in substrate supply and microbial functioning (Kramer and Gleixner, 2008; Hagedorn et al., 2010). As alpine grasslands are replaced by shrubs or trees, relatively labile herbaceous litter and rhizodeposition are increasingly complemented or replaced by more woody, chemically complex inputs and thicker organic layers, which alters the accessibility and quality of C entering the soil (Hagedorn et al., 2019; Speckert et al., 2023). These changes lead to shifts in microbial communities and their functional traits, altering the balance from fast-cycling decomposers adapted to labile inputs towards microbial communities that are better able to process more complex OM (Kramer and Gleixner, 2008; Gunina et al., 2017b; Lu et al., 2017). As substrate quality and microbial community composition strongly regulate enzyme activity, carbon use efficiency, and the priming of native SOM, vegetation-driven changes in plant inputs are likely to influence not only baseline decomposition rates, but also the magnitude and temporal pattern of C losses (Davidson and Janssens, 2006; Bastida et al., 2019; Fanin et al., 2022). Therefore, shifts from pasture to shrub- or forest-dominated vegetation may alter SOM stability both directly, through changed litter inputs, and indirectly, through changes in decomposer communities and their response to warming (Poeplau et al., 2020; Walker et al., 2022).
Rising temperatures increase microbial decomposition of SOM by accelerating microbial and enzymatic processes (Fanin et al., 2022). Temperature increase generally stimulates microbial decomposition of SOM (Davidson and Janssens, 2006) by accelerating microbial and enzymatic processes (Chen et al., 2018). A common first-order approximation is that soil respiration increases by about two- to threefold per 10 °C increase (Davidson and Janssens, 2006), but the realized temperature response is not constant. Instead, it depends on substrate availability (Gershenson et al., 2009), substrate quality (Davidson and Janssens, 2006), and the accessibility of organic matter to decomposers (Moinet et al., 2018). In alpine and subalpine soils in particular, warming responses can be non-linear and may weaken or shift over time as labile C is depleted and microbial respiration acclimates to the prevailing temperature increase (Alster et al., 2023) or community composition shifts toward more heat-adapted taxa (Donhauser et al., 2020). Accordingly, the apparent temperature sensitivity of soil respiration may decline under warming (Abdalla et al., 2024). In decomposition studies under field or lab conditions, warming often causes a strong initial pulse of rapid decomposition (Davidson and Janssens, 2006). This is followed by a decrease of the respiration rate as the labile C pools become depleted or microbial carbon use efficiency (CUE) declines (Davidson and Janssens, 2006; Melillo et al., 2017). Field studies often show a smaller increase compared to laboratory incubation studies under optimized conditions (Hanson et al., 2020; Soong et al., 2021). In alpine and subalpine soils, low baseline temperatures, short growing seasons, and snow cover strongly constrain biological activity (Donhauser and Frey, 2018). Even modest warming can stimulate decomposition and alter microbial substrate use and activity (Streit et al., 2014). In alpine and subalpine grassland soils, warming also increases soil respiration, although its temperature sensitivity may decline under continued warming (Abdalla et al., 2024). In addition, warming-induced reductions in soil aggregation may expose previously protected carbon to mineralization (Poeplau et al., 2020). Overall, rising temperature is likely to accelerate SOM mineralization and efflux of CO2, leading to a positive C–climate feedback (García-Palacios et al., 2021; Chen et al., 2024). Nevertheless, the long-term net effect of warming on C stocks is uncertain, because increased plant growth and C input can partially offset the enhanced SOM decomposition (Chen et al., 2024; Bai et al., 2025). The temperature optimum for microbial processes can vary depending on substrate type, and the sensitivity of different SOC fractions is likely dependent on their quality. Chemically more complex SOM such as lignin may exhibit greater sensitivity compared to more labile compounds (Fierer et al., 2005; Wang et al., 2018). However, some recent studies report similar vulnerabilities of easily decomposable and more complex substances (Ofiti et al., 2021, 2022, 2023; Zosso et al., 2023). Extreme warming may further reduce microbial CUE, as more assimilated C is allocated to maintenance respiration and sensitive taxa experience thermal stress (Nottingham et al., 2022; Dang et al., 2024). In alpine and subalpine ecosystems, compensatory effects may be weaker (Bonfanti et al., 2025). Plant productivity is more limited, and microbial acclimation can be slower, leading to an enhanced vulnerability of soil carbon stocks under climate warming (Qi et al., 2021).
The response of microbial communities in soils to warming is crucial for SOM stability (García-Palacios et al., 2021). Warming can alter microbial community composition and diversity and favour heat-adapted or thermophilic taxa (Melillo et al., 2017; Zosso et al., 2021; Nottingham et al., 2022). Warming is also associated with reduced microbial abundance or biomass (Melillo et al., 2017; Zosso et al., 2021). Such changes in microbial community properties can lower microbial CUE and increase the proportion of C released as CO2 (Domeignoz-Horta et al., 2020). A potential mechanism contributing to these responses is the priming effect (Kuzyakov et al., 2000). Fresh organic C entering the soil can increase the overall activity of soil microorganisms and enable them to decompose native SOM for nutrients and energy. Elevated temperature can increase the strength of positive priming, leading to higher respiration as a result of the decomposition of older SOC (Tao et al., 2024). Warming increases microbial biomass turnover and can promote decomposition of previously stable SOM pools, leading to measurable reductions in soil C stocks on annual to multi-annual timescales (Chen et al., 2024; Tao et al., 2024). However, it still remains uncertain how microbial feedbacks change over time and how microbial communities potentially acclimate to temperature increases and changes in substrate availability and quality.
Long-term experimental data on how temperature and plant-derived C inputs jointly influence soil C dynamics remain scarce (Classen et al., 2015), especially in alpine regions. Recent reviews emphasize that soil microorganisms drive alpine C cycling feedback to climate, but details remain uncertain (Chen et al., 2024; Bai et al., 2025). Thus, there is a need to link temperature regimes, vegetation-type, and substrate dynamics in subalpine soils, and to trace C flow through the soil–microorganism system. Moreover, the combined effects of warming and fresh litter inputs on both short-term decomposition pulses and long-term C stabilization remain poorly quantified in these systems (Tao et al., 2024). To significantly improve our understanding of the vulnerability of SOC in subalpine soils, we conducted a one-year incubation experiment, addressing the following research questions:
-
How do subalpine soils that developed under pasture and forest respond differently to warming and fresh litter inputs?
-
How does temperature affect soil CO2 respiration following fresh litter input?
-
How do functional soil microbial communities respond over time to warming and litter input, and how do these responses relate to changes in carbon mineralization?
We hypothesize the following:
-
Total soil respiration and litter-induced respiration will increase with temperature, with higher peaks and faster declines at warmer temperatures due to rapid depletion of labile carbon. Forest and pasture soils will respond differently due to similar total SOC but contrasting substrate quality and microbial community composition, with forest soils containing more woody-derived organic matter and pasture soils being adapted to steady labile C inputs.
-
Litter addition will stimulate soil respiration and cause positive priming of native SOM, with stronger priming at higher temperatures.
-
Forest soil will show greater CO2 respiration and stronger priming under warming and litter addition than pasture soil, due to differences in substrate quality and microbial community composition, while pasture soil will respond more rapidly but with smaller overall increases.
2.1 Field site and soil sampling
The soil material for the incubation experiment was sampled in July 2020 near Jaun, Canton of Fribourg, Switzerland. The site lies on a south-facing slope at an altitude between 1500 and 1550 m a.s.l. and includes two different vegetation covers, a coniferous forest site and a pasture site (Püntener et al., 2025). The forest site is dominated by Norway spruce (Picea abies L.) with a stand age of at least 130 years. The pasture site is dominated by herbaceous species, mainly ribgrass (Plantago lanceolata L.) and reed fescues (Festuca arundinacea Schreb.) (Speckert et al., 2023). The pasture has been present for at least approximately 160 years, and likely longer, consistent with the long history of pasture use in the region (Hiltbrunner et al., 2013). Annual mean precipitation amounts to 1250 mm and mean air temperatures reach from 0.6 °C in winter to 11.4 °C in summer (Hiltbrunner et al., 2013). The soils were classified as Leptic Eutric Cambisol Clayic (IUSS Working Group WRB, 2015) and developed on a calcareous material (Speckert et al., 2023). The soils are acidic, with only a slight difference in pH between the two sites: pH 5.08 in the pasture soil and pH 4.83 in the forest soil (Püntener et al., 2025). During sampling, the organic layer was removed in the forest soil. In the pasture, the densely rooted mineral top soil (0–5 cm) was also removed before sampling. The mineral soil was sampled at a single location for each site on an area of approximately 1 m2 at a depth of 5–10 cm. In total, approximately 30 kg of soil material were collected at each of the two sites. The material was sieved <2 mm, roots were removed, followed by manual homogenization (Püntener et al., 2025). The soil material was kept for 10 months in a cool, dark place in a storage room in buckets that were loosely covered with aluminium foil allowing air circulation and avoiding complete air drying.
2.2 Incubation setup
The incubation design was intended to improve our mechanistic understanding of SOM decomposition and microbial dynamics in subalpine soils during the growing season and how they respond to future warming. Consequently, seasonal effects as well as daily temperature changes were omitted to simplify the experimental setup. The incubation setup is described in detail in Püntener et al. (2025). Briefly, the two soils were incubated at three different temperatures, with the 2015–2020 average growing season temperature from mid May to mid September of 12.5 °C as lowest temperature treatment and two increased temperature treatments of 16.5 and 20.5 °C. These increased temperature treatments correspond to the expected temperature rise with a high emission scenario (RCP8.5) in Swiss alpine regions by the end of the century (Hock et al., 2019). To trace the decomposition of fresh litter, 13C-labelled – δ13C 2255±248 ‰ Vienna Pee Dee Belemnite (V-PDB) – aboveground plant litter from perennial ryegrass (Lolium perenne) were added to a subset of the samples after a pre-incubation conditioning phase of two weeks. Soil moisture was adjusted to field capacity at the start of the pre-incubation period, checked gravimetrically at least biweekly throughout the incubation experiment and adjusted if necessary. To minimize evaporation and differences in headspace humidity among temperature treatments, vials containing water were placed inside each jar. The incubation was conducted for 360 d, with six destructive soil samplings distributed across the incubation period. This incubation period is much longer than the growing season at the field site, which is the predominant phase of OM decomposition. But to disentangle short-term and long-term SOM decomposition of old SOM and added plant litter, a longer time-span was investigated, which might be equivalent to approximately 3 growing seasons at the field site. We are aware of the artificial length of the experiment and omitted also other fresh OM input such as root exudates and seasonal litterfall to simplify the experimental setup. Respired CO2 was trapped in vials containing NaOH and measured at higher temporal resolution, approximately every 3 d during the initial phase of the incubation and about biweekly during the later experimental stages (sampling scheme in Table S1 in the Supplement).
2.3 Soil respiration: CO2 concentration and C isotope composition
To trace CO2 respired from the soil, a 20 mL sodium hydroxide trap (1 M NaOH) was placed into each incubation jar. The traps were replaced frequently to prevent saturation. The concentration of respired CO2 was estimated using the method by Wollum and Gomez (1970) by measuring the electrical conductivity of the NaOH solution using a conductivity meter (LF 320 Conductivity Meter, WTW, Germany). To correct for temperature, individual temperature measurements were taken for each sample. The measured NaHCO3 in the NaOH traps was converted to CO2 using the calibration by Abiven and Andreoli (2011):
with ECNaOH,sample as the measured conductivity within the NaOH traps (in mS cm−1, corrected to a temperature of 25 °C. From this, total respired CO2–C (CO2–Ctotal; in mg) was calculated using:
with the volume of the NaOH trap vNaOH (in mL) and considering the mass fraction of C (Abiven and Andreoli, 2011; Schiedung et al., 2023).
To measure δ13C of the respired CO2–C, 2.5 mL of each NaOH trap were mixed with 5 mL of 1 M SrCl2 solution to obtain a precipitate of SrCO3 (Schiedung et al., 2023). Each sample was centrifuged (1000 g; 5 min), the supernatant was decanted and the precipitate dried at 50 °C.
After dissolving the precipitate with phosphoric acid on a gas bench (GB), the C isotope composition of the respired CO2 was measured using isotope ratio mass spectrometry (IRMS, Delta V Plus, Thermo Fisher Scientific, Germany). Each sample was injected 10 times and average values were calculated (Breitenbach and Bernasconi, 2011).
All natural abundance isotope ratios are expressed as δ13C relative to the Vienna Pee Dee Belemnite (vs. V-PDB) standard. The labelled samples with litter addition are presented as units of atom % excess (APE), calculated as:
where (atom %) corresponds to the concentration of 13C of the labelled samples and (atom %) to the 13C concentration of the control samples with no litter (Slater et al., 2001).
Priming effect, defined as the change in native SOC mineralization induced by litter addition, was calculated using a two-step isotopic partitioning approach (Kuzyakov et al., 2000). For each sampling day and treatment, the fraction of respired CO2 derived from the labelled litter (fsubstrate) was calculated from the difference in atom % 13C (AP) between samples with litter addition (L+) and control (L−) treatments without litter addition, normalized to the 13C enrichment of the litter:
Native SOC‐derived respiration in litter‐amended samples (Csoil_lbl) was then obtained by:
where Rtotal is the total respiration rate (g C–CO2 kg−1 soil). The priming effect was determined as:
where Rctrl is the native SOC‐derived respiration in the corresponding sample without litter addition. Positive values of PE indicate stimulation of SOC mineralization (positive priming), whereas negative values indicate suppression.
To detect increases in respiration from litter addition, litter-induced respiration (LIR) was calculated as the difference between L+ and L− respiration and thus integrates both litter-derived CO2 and any litter-induced change in native SOC mineralization.
2.4 PLFA analysis
The PLFA analysis was performed using the method described by Frostegård et al. (1991), following the Zosso and Wiesenberg (2021) adaptations of the protocols by Waldrop and Firestone (2006), and Gunina et al. (2017a). For each sample, 4 g of freeze-dried, milled soil material were used for a first extraction for 2 h with 4 mL of extraction solution – of chloroform (CHCl3): methanol (MeOH): citric acid buffer (pH 4) – per g soil. An internal standard (50 µg 1,2-dinonadecanoyl-sn-glycero-3-phosphocholine; PC19:0; Avanti Polar Lipids, USA) was added for quantification. After centrifuging for 10 min at 800 g, the supernatant was transferred to separation funnels. The extraction was repeated three times with 10 mL of the solvent mixture during each round. After phase separation, the organic phase was eluted and reduced to 100 µL using a multivapor (Multivapor P-6, Büchi Labortechnik AG, Switzerland). The individual fractions were separated using a column with activated silica gel (Silica 60, Honeywell Fluka, USA; activated at 110 °C overnight). The neutral fraction was eluted with 5×1 mL CHCl3, the glycolipid fraction with 4×5 mL acetone, and the phospholipid fraction with 4×5 mL MeOH. After reduction to 100 µL, remaining water was removed using a column filled with anhydrous Na2SO4. The method by Wiesenberg and Gocke (2017) was used for methylation. Briefly, 5 µg of D39C20 acid were added as a control standard, followed by dissolving in 300 µL dichloromethane (CH2Cl2). As methylation reagent, 500 µL of a BF3–MeOH solution (10 % , Sigma Aldrich, Inc., USA) were added to each sample. The samples were heated at 60 °C for 15 min on a heating block. After the samples reached room temperature, 500 µL of ultra-purified water was added. The samples were centrifuged, and the organic phase was transferred onto anhydrous Na2SO4, filtered, and the filtrate collected in an autosampler vial. CH2Cl2 was added another 5–8 times to the methylation solution until the organic phase was colorless.
Quantification was carried out using a gas chromatograph with a flame ionization detector (GC-FID, Agilent 7890 B, Agilent Technologies, Inc., USA, equipped with 50 m × 0.2 mm × 0.32 µm Agilent J&W DB-5MS column) with a multi-mode inlet (MMI). The GC temperature program was as follows: Start at 50 °C for 4 min, increase to 150 °C with a rate of 10 °C min−1, followed by an incremental increase (2 °C min−1 to 160 °C, 0.5 °C min−1 to 170 °C, 0.2 °C min−1 to 190 °C, 2 °C min−1 to 210 °C) to a maximum temperature of 320 °C that was held for 15 min (Zosso and Wiesenberg, 2021). Compound peaks were matched against a suite of 24 fatty acid standards (Larodan; Sigma-Aldrich; Avanti Polar Lipids) and additionally confirmed by running samples on a GC (Agilent 6890 N, Agilent Technologies, Inc., USA, equipped with the same column as the GC-FID) coupled to a mass spectrometer (MS, Agilent 5973 N, Agilent Technologies, Inc., USA). The spectra were also compared to Wiley/NIST mass spectra libraries.
The PLFAs were grouped as in Zosso and Wiesenberg (2021) according to Willers et al. (2015) to differentiate the functionally different parts of the microbial communities: fungi (C), Gram negative bacteria (Gram−; C16:1ω5c, C16:1ω7c, C16:1ω9c, C18:1ω5c, C18:1ω11c), Gram positive bacteria (Gram+; iC14:0, aC14:0, iC15:0, aC15:0, iC16:0, aC16:0, aC17:0), actinobacteria (10MeC16:0, 10MeC18:0), and cyclopropyl bacteria (cyC17:0, cyC19:0). Microbial abundance was calculated using the sum of these diagnostic PLFA markers and the non-diagnostic saturated PLFAs (C14:0, C15:0, C16:0, C17:0, C18:0), which are general bacterial markers (Zosso and Wiesenberg, 2021). The total concentrations of PLFAs were calculated in relation to the internal standard.
Carbon isotope composition of the PLFAs was analyzed using a GC (TRACE 1310, Thermo Fisher Scientific, Germany) which is coupled to an isotope ratio MS (Delta V Plus IRMS, Thermo Fisher Scientific, Germany) via GC-Isolink II and ConFlo IV (Thermo Fisher Scientific, Germany). For compound-specific isotope analysis of phospholipid-derived fatty acids (PLFAs), the isotopic effect of adding methyl groups during BF3–MeOH methylation was corrected using a mass balance approach adapted from Dignac et al. (2005) (Eq. 4):
In this equation, δUD denotes the carbon isotopic ratio of the original, underivatized PLFA; nD is the total number of carbon atoms in the methylated PLFA (FAME); nUD is the number of carbon atoms in the underivatized fatty acid; δD is the isotopic ratio of the methylated PLFA measured by GC-IRMS; nMeOH represents the number of carbon atoms introduced from the methanol reagent during derivatization (one carbon per methyl group); and δMeOH is the carbon isotopic ratio of the methanol reagent, measured via GC-IRMS relative to underivatized reference compounds.
Consistent with the reporting of the C isotope composition of the respired CO2, δ13C values of samples without litter addition are expressed relative to the V-PDB standard, whereas samples with 13C-labelled litter are reported as atom % excess (APE).
Only PLFA biomarkers representing the functional groups actinobacteria, general bacteria, Gram+ bacteria, and Gram− bacteria are reported here, as clear and quantifiable peaks were obtained only for these compounds.
2.5 Statistics
All statistical analyses were conducted in RStudio (version 2025.05.0+496, using R version 4.4.1, R Core Team, 2025). Respiration and isotope data were tested for normality (Shapiro–Wilk) and log-transformed. For visualization and descriptive summaries, we calculated treatment-wise means and standard deviations (SD) of respiration rates, cumulative respiration and C isotope composition. To assess the treatment effects over the incubation time, raw respiration values were analyzed using linear mixed-effects models (LMMs) with the lmer() function from the lme4 package. Temperature, vegetation type (forest vs. pasture), litter addition (L+ vs. L−), and sampling day were included as fixed effects. Sample number was treated as a random intercept to account for the repeated measurements of the same incubation jars during the incubation. For pairwise post-hoc comparisons, we calculated marginal means (EMMs) using the emmeans package. Differences between temperature levels, vegetation types, and litter treatments were tested using a Tukey-adjusted pairwise comparison.
3.1 Soil respiration
Cumulative respiration increased with increased temperatures T16.5 and T20.5 compared to control temperature T12.5 (both p<0.001) for both, L− (Fig. 1a) and L+ soil samples (Fig. 1b). Litter addition significantly increased the respiration rate (p<0.001), with the strongest responses observed under elevated temperature. Slightly higher respiration rates were measured for forest samples compared to pasture samples in both, L− and L+ soils, especially at higher temperatures. However, the difference between the two vegetation types was not significant (p=0.45). The respiration trajectories between L− and L+ measurements differed (p<0.001). In L+ treatments, early respiration (within the first 30 d) accounted for 83 %–90 % of total cumulative respiration across all temperatures and vegetation types. In contrast, L− treatments showed substantially lower early respiration, with averages ranging from 58 % to 74 %, indicating a more gradual release of CO2 over time.
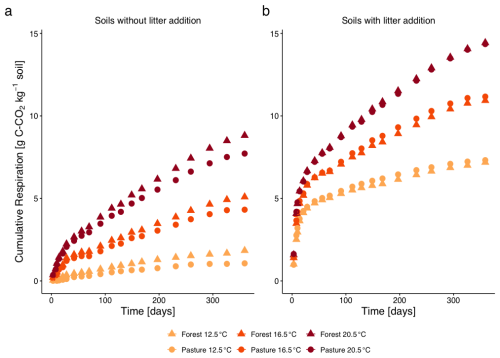
Figure 1Cumulative CO2–C respiration (mean ± SE, errors smaller than symbols and thus not visible) from subalpine forest and pasture soils without litter addition (a) and with 13C-labelled aboveground L. perenne litter addition (b) over 360 d of incubation at three temperatures (12.5, 16.5, 20.5 °C). Different colours represent the different incubation temperatures, different symbols the two different land cover types.
The initial increase of the litter-induced respiration (LIR, Fig. 2a) with a maximum on day 8 exhibited a clear increase with increasing temperature (on day 3 and day 8, all p<0.0001). Additionally, the respiration was between 4 %–23 % higher in the pasture soil compared to the forest soil (day 8, all p<0.0001). After the initial peak, LIR decreased drastically and faster with higher temperature, and faster for pasture than for forest samples (day 10: p<0.0001 for temperature and land use). For the remaining incubation duration, LIR remained at a low rate, however with small increases at days 91 and 231 for all treatments. In forest T12.5, LIR dropped by more than 67 % from day 8 to day 10 (p<0.0001), followed by a partial rebound of 36 % by day 14. Smaller secondary peaks of >30 % also occurred in forest T20.5 (e.g., between day 91 and 112, p<0.05). A deviation was observable from day 70 with the highest respiration for pasture T16.5, which became even more pronounced on day 91 (p<0.04) and remained highest until day 197. This increased respiration was also reflected in the cumulative LIR, which was highest for pasture T16.5 (Fig. 2b), being 9.6 % higher relative to T12.5 compared to only 6.2 % higher LIR at T20.5. For the overall incubation period, higher cumulative LIR was observed for pasture samples compared to forest samples (p<0.0001). Identical as for pasture soil, cumulative LIR in the forest soil was higher for samples with T16.5 (9.3 % higher than T12.5), followed by T20.5 (5.2 % higher than T12.5).
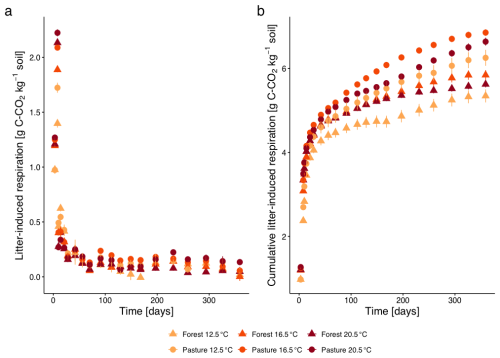
Figure 2(a) Litter-induced respiration (LIR; mean ± SE), calculated as the difference between respiration of soils with and without L. perenne litter addition, for subalpine forest and pasture soils over the incubation period at three temperatures (12.5, 16.5, 20.5 °C). Different colours represent the different incubation temperatures, different symbols the two different land cover types. (b) Cumulative LIR (mean ± SE) over 360 d.
3.2 δ13C of respired CO2
The C isotope composition of the respired CO2 of the L− soils only showed small temporal fluctuations over the whole incubation period (Fig. 3a). At the beginning of the incubation, we observed a shift towards increasing δ13C values in both, forest and pasture soils. This was followed by a shift to more negative values with minima around days 56 to 70, which were more pronounced in forest soil than in pasture soil. From there, δ13C increased until day 197, followed by a decrease and values becoming more negative towards the end of the incubation experiment. In warmed samples, the shifts occurred slightly faster (p=0.002) compared to T12.5 samples. δ13C dynamics did not differ between forest and pasture soils.
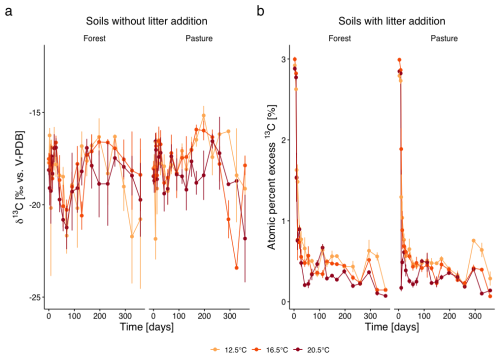
Figure 3(a) δ13C values (‰ vs. V-PDB; mean ± SD) of the respired CO2 from subalpine forest and pasture soils without litter addition over the incubation period of 360 d under three incubation temperatures (12.5, 16.5, 20.5 °C). Different colours represent the different incubation temperatures, different symbols the two different land cover types. (b) Atom % excess (APE; mean ± SE) 13C of respired CO2 from soils with L. perenne litter addition over 360 d.
Litter addition significantly increased 13C concentration (p<0.0001) in both forest and pasture soils, indicated by the high atom percent excess of 13C (APE 13C) at the beginning of the incubation experiment (Fig. 3b). At day 3, APE 13C reached about +3.00 % in forest soils and +2.85 % in pasture soils, corresponding to relative increases of +275 % and +261 % over control samples. The 13C excess decreased rapidly within the first two weeks of the incubation experiment. For the remaining incubation period, APE 13C only decreased slowly, with some fluctuations throughout. Warming significantly modulated this decrease (p=0.02), which was stronger in the beginning and a consequential lower excess throughout the incubation period. No significant difference was detectable between forest and pasture soils.
3.3 Priming
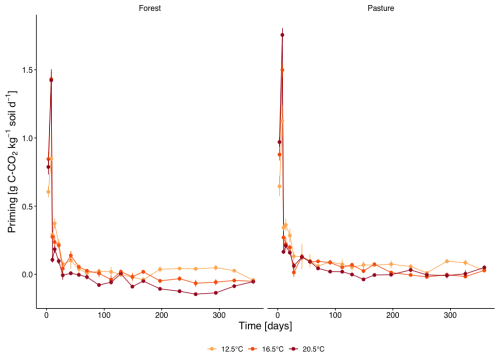
Priming of native soil C (Fig. 4) increased in the first week of incubation for both forest and pasture with a peak on day 8 with stronger priming with higher temperature (forest: T12.5 0.84±0.45 g C-CO2 kg−1 soil d−1, T16.5 1.35±0.71 g C-CO2 kg−1 soil d−1 and T20.5 1.42±0.79 g C-CO2 kg−1 soil d−1; pasture: T12.5 1.15±0.52 g C-CO2 kg−1 soil d−1, T16.5 1.44±0.76 g C-CO2 kg−1 soil d−1 and T20.5 1.66±0.79 g C-CO2 kg−1 soil d−1). Subsequently, priming declined rapidly to near zero by day 28 and remained low thereafter. A small, but significant overall higher priming was measured in pasture versus forest soil (p=0.048). No significant overall differences in priming were detectable between different temperature treatments, but the warmed treatments exhibited a faster drop of priming rates compared to T12.5 between days 10–21 (p<0.01).
3.4 Phospholipid fatty acid composition and compound-specific isotopes
In the beginning of the incubation experiment, phospholipid fatty acid (PLFA) concentrations in soils without litter addition (L−) ranged between approximately 300 and 550 µg g−1 dry soil. Across all treatments, total PLFA concentrations declined over time. PLFA concentrations in L− samples (Fig. 5a and b, relative abundance Fig. S1a and b in the Supplement) at T20.5 were lower than those at T12.5 and T16.5, especially at later stages of the experiment.
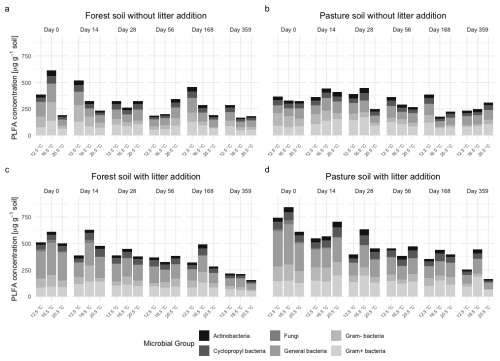
Figure 5Total phospholipid fatty acid (PLFA) concentrations for functional microbial groups (general bacteria, Gram+ bacteria, Gram− bacteria, fungi, actinobacteria, cyclopropyl bacteria) in (a) subalpine forest soil without litter addition, (b) subalpine pasture soil without litter addition, (c) subalpine forest soil with L. perenne litter addition, and (d) subalpine pasture soil with L. perenne litter addition. Soils were incubated for 360 d at three temperatures (12.5, 16.5, 20.5 °C).
In forest L− soil (Fig. 5a), Gram+ and Gram− bacteria consistently accounted for up to 60 % of total PLFA at all timepoints (Fig. S1a). Cyclopropyl and general bacteria proportions stayed relatively constant during the incubation, although with small increases of cyclopropyl bacteria at T20.5 during the middle of the incubation period at days 56 and 168. Fungal PLFA concentrations remained low throughout the incubation, with values rarely exceeding 25 µg g−1 and a slight increase at the beginning of the incubation (day 14 to 28).
In pasture L− soil (Figs. 5b and S1b), the microbial composition was also dominated by Gram+ and Gram− bacteria, which declined steadily over time, with a stronger decrease for T20.5 compared to the lower temperature treatments. The same trend was visible for the general bacteria group. The abundance of fungi was low (around 10–20 µg g−1 on average), while actinobacteria were slightly higher (15–30 µg g−1), but both groups remained relatively constant throughout the incubation period with only small fluctuations.
Litter addition (L+) led to a strong increase in PLFA abundance at the beginning of the incubation period, exceeding 700 µg g−1 dry soil. Throughout the incubation period, these high values decreased to levels approaching or below those of L− samples. Interestingly, highest total PLFA concentrations were most often found at T16.5 across all timepoints.
Forest L+ (Figs. 5c and S1c) had highest PLFA general bacteria group concentrations during early incubation, especially at T12.5 and T16.5. General bacteria, Gram+, and Gram− bacteria concentrations declined strongly over time, with a stronger decrease under elevated temperature. Fungal PLFAs showed an increase in the beginning of the incubation experiment, with a rapid decrease after the first 28 d and they were almost absent by the end of the incubation experiment. Similar to L− soils, cyclopropyl bacteria exhibited an increase at days 56 and 168 under elevated temperatures.
Pasture L+ (Figs. 5d and S1d) showed a similar pattern to the forest soil, with general bacteria as dominating microbial group and a fast decline of PLFA concentrations in the first 28 d, especially under elevated temperature. At intermediate timepoints (days 56 and 168) in the warmed treatments (T16.5 and T20.5), cyclopropyl bacteria increased more strongly than in L− soils, where this mid-incubation increase was less pronounced. In comparison to pasture L−, Gram+ and Gram− bacteria were less dominant and exhibited a stronger decrease with warming. Fungal PLFAs showed a short-lived increase between days 14 and 28, especially at T12.5 and T16.5, but declined quickly thereafter. Actinobacteria remained a minor group (typically 10–25 µg g−1), but exhibited slight increases at days 56 and 168, especially under cooler temperatures (T12.5 and T16.5).
In L− soils (Fig. 6a), weighted average δ13C values of all microbial functional groups based on PLFA remained near natural abundance throughout the whole incubation period. The δ13C values of the individual groups varied between −32 ‰ to −26 ‰, with general and Gram+ bacteria showing values closer to −28 ‰ to −30 ‰, and Gram− bacteria occasionally reaching −26 ‰. The temporal variation was only minor but still visible for some groups, with fluctuations of up to ±2.5 ‰ observed at days 28, 56, and 168. The differences between the individual functional groups were small, with pasture soils being slightly more depleted in 13C than forest soils, with a shift of ca. 1 ‰–22 ‰.
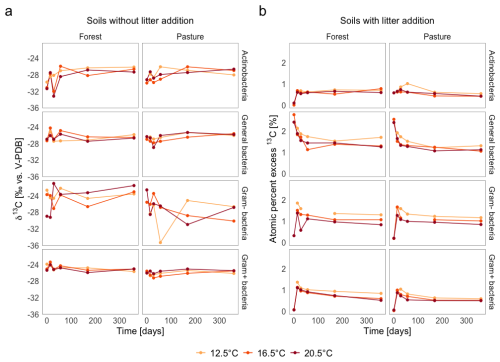
Figure 6Mass-normalized weighted average δ13C (‰ V-PDB for soils without litter addition; atom % excess for soils with litter addition) of PLFA biomarkers representing functional microbial groups in (a) soils without litter addition and (b) soils with litter addition over 360 d at three incubation temperatures.
In L+ soils (Fig. 6b), the PLFA biomarkers showed a clear incorporation of the 13C labelled litter C. This was evident from strongly elevated δ13C values (e.g., up to −10 ‰ in general bacteria at day 14) compared to natural abundance, which remained below −25 ‰. 13C excess peaked at day 14. Only the general bacteria group exhibited the highest values at the beginning of the incubation period. Following the initial peak, the 13C label decreased in all groups rapidly, but remained above natural abundance during the whole incubation period. For the other microbial functional groups (Gram+ and Gram− bacteria, actinobacteria), 13C incorporation was highest at day 14 or 28, but the peak was less pronounced than for general bacteria. Across all time points, a temperature effect was visible, with the highest 13C excess values generally found at T12.5, while values at T16.5 and T20.5 were more similar and clearly lower.
4.1 Elevated temperature stimulates subalpine soil organic matter decomposition
Our one-year incubation experiment demonstrated that higher temperature accelerated the decomposition of organic matter in the subalpine soils investigated here, which is in line with other studies identifying temperature as an important regulator of C turnover in soils (Davidson and Janssens, 2006; Nottingham et al., 2019). Total cumulative CO2 emissions were significantly higher at higher temperatures compared to the current growing season temperature of 12.5 °C, indicating a stronger microbial breakdown of both, native SOM and the added litter under increased temperature. These findings confirm our hypothesis and align with the positive relationship between temperature and SOM mineralization rates (Crowther et al., 2016; Chen et al., 2024). Higher temperatures increase microbial metabolic rates (Xu et al., 2023) and enzyme activities (Chen et al., 2018), which leads to increased decomposition of SOM and thus an increased release of CO2 from the soils. This observed increase argues for a positive soil carbon–climate feedback, especially in these high-altitude soils that are rich in SOC. Already the lower temperature increase of +4 °C increased total CO2 efflux up to 50 % compared to control temperature. This is consistent in direction with several field studies (Hanson et al., 2020; Soong et al., 2021; Chen et al., 2024), where a temperature increase led to an increased heterotrophic respiration of 26 %–37 % compared to our 50 %–100 % increase under optimal conditions for higher temperature. This temperature-driven increase of CO2 flux emphasizes the vulnerability of alpine and subalpine soil SOC stocks to even modest warming expected with climate change (Crowther et al., 2016; Wang et al., 2022).
The warming effects were more pronounced in the litter-amended soils for both forest and pasture. While Bastida et al. (2019) revealed in a global data analysis that fresh labile C inputs can stimulate microbial activity and modulate the priming responses, our results extend this pattern to subalpine soils. This aligns with experiments in alpine regions showing that warming elevates microbial activity and substrate use and that greater fresh inputs intensify warming-induced soil C losses in alpine ecosystems (Streit et al., 2014; Walker et al., 2022; Ye et al., 2022). This was also visible in the incubated soils for total SOM and the less decomposable lignin monomers, where decomposition was only higher with higher temperatures in the presence of fresh litter (Püntener et al., 2025). One reason for this might be positive priming (Kuzyakov, 2010). Addition of labile C in the form of the litter accelerated the decomposition of native SOM. The metabolic cost of warming is therefore offset by this improved and increased energy supply (Wild et al., 2016). This higher decomposition of native SOM, which consists of older, less easily decomposable organic compounds, aligns with studies reporting that these pools can be particularly responsive to increased temperatures (Craine et al., 2010; Frey et al., 2013), especially with a high abundance of labile carbon (Püntener et al., 2025).
The priming pattern indicates that this stimulation of native SOM mineralization was highly transient. Priming peaked during the first week, when litter-induced respiration was the highest, and declined to near zero by day 28. This temporal coupling suggests that the litter pulse initially activated microbial populations that are able to exploit both the added substrate and a fraction of native SOM. However, this effect weakened rapidly once the most accessible litter-derived compounds were depleted and microbial demand shifted (Kuzyakov et al., 2000; Kuzyakov, 2010; Bastida et al., 2019; Tao et al., 2024). The faster decrease of priming in the warmed treatments is consistent with the more rapid decrease in 13C excess and the steeper post-peak decline in litter-induced respiration, indicating that higher temperature accelerated the turnover of the labile litter fraction and shortened the period during which fresh inputs stimulated native SOM mineralization (Schindlbacher et al., 2011; Wild et al., 2016; Pold et al., 2020). Thus, litter addition clearly stimulated respiration and induced positive priming, confirming the second hypothesis, whereas the temperature effect on priming was expressed mainly in the early phase of decomposition rather than as a sustained increase over the full incubation period. Likewise, litter-induced respiration followed the predicted pattern of a stronger early peak and faster decline at higher temperature, although cumulative LIR was highest at 16.5 °C rather than at 20.5 °C. These contrasts are consistent with differences in vegetation history and associated microbial communities, but because the comparison is based on one forest and one pasture site, site-specific properties cannot be fully separated from vegetation effects.
4.2 Contrasting decomposition dynamics between pasture and forest soils
During the first week of incubation, litter-induced respiration (LIR) showed marked differences in peak timing and magnitude between the soils. The pasture soil responded more rapidly to the litter input than the forest soil. Litter-induced respiration peaked during the first week of the incubation for both soils, but was significantly higher in pasture soils. The decrease after the first peak was faster in pasture than forest soils, which was also identical to the δ13C decrease, reflecting a rapid mineralization of the added plant litter. In soils without litter addition, the δ13C signal of respired CO2 followed a similar overall pattern in both soils, with values becoming less negative at the start of the incubation and later shifting back toward more negative values. The initial shift occurred only slightly earlier in the pasture (around day 3–10) than in the forest soil (around day 10), however the shift back to more negative values happened earlier in pasture soil after around 20 d, whereas in the forest, the minimum of that shift was only reached after around 80 d. Consistently, PLFA-derived microbial biomass peaked earlier and stronger in the pasture soils, whereas forest biomass peaked later and less strongly. These differences in the early stage of the incubation period are likely due to differences in microbial community composition. The pasture site supposedly experienced greater daily temperature shifts and a higher concentration of readily available C input from grass litter and root exudates (Peplau et al., 2023). These systems often harbor greater proportions of fast-growing, copiotrophic microorganisms than, e.g., forest soils (Solly et al., 2014; Liu et al., 2023). In our data, this is supported by the larger concentration of Gram− in pasture soils, which are known to mainly process more labile C sources (Creamer et al., 2016; Zheng et al., 2021). In the pasture soil, the δ13C value of the respired CO2 also exhibited fluctuations earlier in the incubation period, indicating a rapid shift in substrate use from one C source to another, a pattern consistent with studies showing that grassland microbial communities can rapidly adjust their metabolism to changes in available substrates (Creamer et al., 2016; Liu et al., 2023; Wang et al., 2023). This faster response has also been observed in alpine grasslands where microbial communities are dominated by fast-growing taxa (Budge et al., 2011; Donhauser et al., 2020). The forest site, in contrast to the pasture soil, is characterised by a microbial community that is more adapted to the litter input from spruce containing more polyphenols and lignin, which are harder to decompose than grass litter in the pasture soil (Solly et al., 2014; Ortiz et al., 2016). For our soils, this difference in the microbial community is documented by the larger concentration of Gram+ bacteria and fungi in the forest soil compared to the pasture soil. Certain members of these functional microbial groups (e.g. firmicutes) are known to process preferentially less decomposable organic compounds (Kramer and Gleixner, 2008; Zhang et al., 2016; Lu et al., 2017). In the forest soil, the δ13C trends shifted more slowly, which may reflect a more buffered system with microbial communities adapted to the continuous but lower-quality litter inputs from spruce needles (e.g., high in lignin) (Kramer and Gleixner, 2008; Hiltbrunner et al., 2013; Lu et al., 2017). Such microbial communities may require more time to switch to alternative carbon sources, leading to a delayed response of the isotopic signal, as reported for forest soils with more complex SOM pools and microbial communities specialized in decomposing this type of OM (Schindlbacher et al., 2011; Ortiz et al., 2016; Gunina et al., 2017b). In our experiment, the presence of Lolium perenne litter in both soils acted as a high-quality substrate and levelled out possible substrate differences. Nevertheless, the pasture soil might have had a slight advantage, being adapted to grass litter inputs and therefore rapidly decomposing the added ryegrass, whereas the forest microbial community was initially less efficient in decomposing the less familiar substrate.
The temporal pattern of native SOM decomposition can also further support the theory of the two different systems. The initial stimulation of native C mineralization was stronger in pasture than in forest soils but declined more rapidly. Such pronounced, short-lived responses are characteristic for a system that is dominated by a copiotrophic microbial community (Bastida et al., 2019; Wang et al., 2023). The priming response therefore followed the same general pattern as litter-derived respiration and microbial biomass development: Pasture soil exhibited a stronger and earlier pulse, whereas forest soil responded more gradually and over a longer period. This indicates that the land-cover contrast was expressed mainly in the kinetics of decomposition and priming rather than in a consistently larger cumulative response in forest soil. In this respect, the third hypothesis was only partly confirmed: pasture soil indeed responded more rapidly, but stronger priming was not observed in forest soil. Instead, priming was slightly but significantly higher in pasture soil overall, consistent with a more copiotrophic microbial community rapidly exploiting fresh inputs (Creamer et al., 2016; Bastida et al., 2019; Wang et al., 2023).
Together, the findings of our experiment characterise the pasture soil as a dynamic, fast cycling system, in contrast to a more static, slow cycling system in the forest soil. This has important implications for C cycling in alpine and subalpine soils under the influence of climate change. Rising temperatures might lead to a faster loss of C from dynamic systems like pasture, while slower cycling systems like our forest soil might temporarily mitigate C losses (Verbrigghe et al., 2022; Peplau et al., 2023; Chen et al., 2024). However, this does not guarantee long-term stability of SOC in a warmer future, as we have seen in our L− cumulative respiration rate, which was higher for the forest than for the pasture soil, indicating an additional strong decomposition in the absence of fresh litter. Additionally, a slow adaptation to higher temperature, as happens with climate change, will lead to shifts in microbial communities, reducing the soils' resistance to loss of SOC (Nottingham et al., 2019; Yuan et al., 2021).
4.3 Diverging temperature optima of total and litter-induced respiration
In contrast to the subsequent rise of total soil respiration with temperature, LIR peaked at the intermediate temperature of 16.5 °C. This apparent contradiction highlights distinct temperature sensitivities of microbial communities when decomposing native SOM versus the added litter. The LIR optimum was consistent across both soils from different vegetation covers and coincided with the highest PLFA concentrations, suggesting that the microorganisms that decompose the litter operate near their physiological optimum at this temperature (Kravchenko et al., 2019).
At highest temperature T20.5, the lower microbial biomass, a faster loss of the 13C label, and an increase in cyclopropyl PLFA point to a microbial response in which direct temperature stress and accelerated depletion of readily available litter compounds likely acted together (Willers et al., 2015; Zhu et al., 2023). Elevated temperature may have increased microbial maintenance demands and reduced growth efficiency (Schindlbacher et al., 2011; Li et al., 2019), while also accelerating turnover of labile C (Djukic et al., 2013). This suggests that the communities shifted earlier from biomass production to maintenance-dominated metabolism, consistent with reduced microbial CUE (Schindlbacher et al., 2011). Accordingly, lower growth efficiency at elevated temperature likely promoted faster litter decomposition while limiting microbial biomass accumulation (Frey et al., 2013; Pold et al., 2020; Dang et al., 2024). This shift in C partitioning at higher temperature likely shortened the duration of high litter decomposition rates at T20.5.
Presumably, litter use dynamics, transient priming, and microbial physiology together explain the divergence in thermal optima. Litter-derived OM consists to a large part of simple, more labile compounds that can be rapidly decomposed by fast-growing microbial taxa, especially Gram− bacteria (Creamer et al., 2016; Zheng et al., 2021). These microorganisms may have a narrower thermal tolerance range and a lower heat resistance than microbial groups involved in the decomposition of SOM, such as certain Gram+ bacteria or fungi (Cui et al., 2022; Zhu et al., 2023). With the highest temperature T20.5 likely exceeding their thermal optimum, the litter decomposing microorganisms may lose competitive advantage, resulting in a reduced LIR even though total soil respiration remains high due to SOM decomposition by more thermotolerant taxa.
This also helps explain the priming dynamics. Highest priming coincided with the phase of strongest litter-derived respiration and elevated microbial biomass in the litter-amended treatments, indicating a close link between early litter processing and stimulation of native SOM mineralization (Wild et al., 2016). As PLFA concentrations declined over time, and particularly as cyclopropyl markers increased at the highest temperature, priming also weakened, consistent with a shift from rapid substrate exploitation to lower biomass and increased physiological stress (Willers et al., 2015; Zhu et al., 2023). Another influence is the thermal adaptation of microbial communities. Laboratory warming experiments have shown that the microbial growth temperature optimum can increase after prolonged exposure to high temperatures, however these shifts are often accompanied by community changes towards more heat-adapted taxa (Donhauser et al., 2020). In our controlled incubation, the absence of such long-term adaptation processes may explain why LIR peaked below the highest temperature tested. In natural field settings, such adaptations could be slower or incomplete, particularly in alpine and subalpine soils with historically low temperatures.
These results imply that standard incubation temperatures of 20–25 °C may exceed the natural optima for litter decomposition in these subalpine soils, especially for those parts of the microbial community that are adapted to cooler conditions. Applying local temperature ranges in laboratory incubations would better reflect the in situ microbial performance and capture realistic temperature responses. The observed difference between LIR and total respiration also underscores that litter-derived and SOM-derived CO2 fluxes cannot be assumed to respond identically to warming; fresh inputs may have narrower, lower-temperature optima shaped by substrate traits and decomposer ecology (Moinet et al., 2018). Such differences in thermal optima among carbon pools should be considered when projecting soil carbon–climate feedbacks, as warming may shift the relative contribution of litter versus SOM to total CO2 respiration.
Accordingly, the first hypothesis was supported for the stronger early respiratory response to litter addition and its faster decline at higher temperature, but only partly supported for cumulative LIR, which was maximal at intermediate rather than highest temperature.
4.4 Broader implications
Our findings provide new insights into the response of subalpine soils to climate warming and vegetation shifts and the resulting consequences for SOC stability and ecosystem functioning across alpine landscapes (Donhauser et al., 2020). The observed increase of total soil respiration under elevated temperature, combined with a substrate-specific decomposition response and contrasting microbial dynamics between the two studied soils, suggest that subalpine and alpine soils are vulnerable to enhanced C losses under future climatic conditions.
While the temperature treatments were chosen around the current growing-season temperature and plausible warming levels, the present incubation primarily resolves process-level responses during biologically active phases. The constant temperature design is particularly useful for disentangling the direct effects of warming and fresh litter inputs on respiration, priming, and microbial dynamics (Chen et al., 2023), without the additional complexity introduced by fluctuating thermal regimes (Adekanmbi et al., 2022). In subalpine soils, snow cover buffers winter soil temperatures and contributes to seasonal patterns of microbial activity and CO2 exchange (Merbold et al., 2013; Gavazov et al., 2017), while snowmelt can trigger abrupt shifts in microbial functioning and biogeochemical cycling (Broadbent et al., 2021; Rindt et al., 2023). Our results are therefore most informative for identifying the direction and relative strength of warming effects during active decomposition: Warming increased the potential for SOM mineralization, accelerated the initial turnover of fresh litter, and intensified a short-lived priming response.
The different temperature sensitivities between native SOM and litter decomposition in our incubation experiment are particularly relevant, as they imply that warming may alter the relative contribution of different C pools to microbial soil respiration. Particularly, a potential long-term depletion of native SOC stocks could be the result of a continued SOM mineralization at high temperatures (Davidson and Janssens, 2006; Nyberg and Hovenden, 2020). Previous studies have shown that warming can reduce microbial C retention (Frey et al., 2013; Domeignoz-Horta et al., 2020), thereby increasing the proportion of assimilated carbon lost as CO2 rather than incorporated into the microbial biomass. These observed trends suggest that warming could shift alpine and subalpine soils toward increased C vulnerability, especially during seasons or in ecosystems where the availability of litter input is limited.
The transient priming pulse observed after litter addition further suggests that fresh organic inputs can enhance C loss not only through decomposition of the added substrate itself, but also through a short-lived stimulation of native SOM mineralization (Kuzyakov et al., 2000; Tao et al., 2024). In our experiment, this effect was most pronounced during the early phase of litter processing, when respiration rates and microbial biomass were highest, and weakened later as microbial biomass declined and the 13C excess of respired CO2 in the litter-amended treatments decreased.
Changes in the vegetation composition such as treeline upward movement and shrub encroachment or afforestation, which are widespread in alpine regions, can lead to further changes in SOC dynamics (Dengzeng et al., 2022; Laorden-Camacho et al., 2025). The results of our incubation experiment show that the two studied soils differed in their decomposition dynamics, even under identical experimental conditions. Similar differences have been found in other alpine ecosystems, where shrub encroachment changed the microbial community composition and SOC dynamics (Dengzeng et al., 2022; Laorden-Camacho et al., 2025). This supports that changes in litter quality and microbial community composition induced by changing vegetation can influence the turnover of SOM under warming.
Furthermore, our findings reveal that warming increased respiration in the soils without litter addition, indicating enhanced microbial mineralization of native SOC in the absence of fresh litter inputs (Nyberg and Hovenden, 2020). This higher CO2 efflux suggests that warming can accelerate the breakdown of SOM that would otherwise remain more stable under cooler conditions, potentially by increasing microbial activity and enzymatic degradation rates, and by reducing physical or chemical protection of organic matter (Ofiti et al., 2022; Zosso et al., 2023).
Combining all these dynamics, the pasture soil studied here responded to warming with larger, more transient CO2 fluxes, whereas the forest soil showed a slower, more sustained SOM loss. More generally, these differences are consistent with vegetation-related contrasts in subalpine soils, but they should be interpreted cautiously because vegetation cover and site identity were not independently replicated in this study. These responses are not only shaped by temperature, but also by the composition of soil microbial communities induced by vegetation shifts and the availability and quality of litter input.
Afforestation, cessation of grazing of alpine pastures followed by shrub encroachment or shrub clearing can have a strong influence on soil C dynamics as our findings emphasized. These interventions must therefore be evaluated not only for their aboveground effects but also and likely even more for their influence on SOC dynamics. Warming-induced increases in SOM mineralization might negate any anticipated increase of SOC concentration due to changes in land-use and vegetation cover, specifically in such alpine soils that are comparatively enriched in SOC (Speckert et al., 2023). Overall, the data confirm a warming-induced increase in total soil respiration and a clear litter-triggered positive priming pulse, but they also show that both responses depend strongly on timing and vegetation history. Warming strengthened the initial priming pulse but also accelerated its decline, and the strongest short-term priming response occurred in pasture rather than forest soil.
Under future warming, alpine and subalpine soils are at risk of becoming significant sources of atmospheric carbon. Our findings of a one-year laboratory incubation experiment demonstrate that not only temperature but also vegetation type and microbial functioning critically shape soil carbon dynamics. The strong stimulation of CO2 release with warming highlights the sensitivity of native SOM to even moderate temperature increases. Distinct decomposition patterns in pasture and forest soils show that microbial communities and their vegetation history influence how quickly and persistently soil carbon is lost. Importantly, the non-linear response of litter-induced respiration, with a peak at intermediate temperatures, suggests that microbial efficiency declines beyond certain thresholds, thus limiting their capacity to retain carbon under long-term warming. These mechanisms together point to a destabilization of alpine soil organic carbon stocks under projected climate scenarios. To anticipate long-term consequences, it will be essential to integrate microbial thermal responses and vegetation shifts into ecosystem models and monitoring efforts.
The code used in this study is available from the corresponding author upon request.
The data for this study can be accessed from https://doi.org/10.5281/zenodo.20094476 (Püntener et al., 2026).
The supplement related to this article is available online at https://doi.org/10.5194/soil-12-599-2026-supplement.
DP: conceptualization, data curation, formal analysis, investigation, visualization, supervision, writing – original draft, writing – review & editing. PZ: investigation, data curation, formal analysis. TCS: conceptualization, investigation, data curation, supervision. CLT: conceptualization, investigation, data curation, writing – review & editing. GLBW: conceptualization, funding acquisition, investigation, methodology, project administration, resources, supervision, writing – review & editing.
The contact author has declared that none of the authors has any competing interests.
Publisher's note: Copernicus Publications remains neutral with regard to jurisdictional claims made in the text, published maps, institutional affiliations, or any other geographical representation in this paper. The authors bear the ultimate responsibility for providing appropriate place names. Views expressed in the text are those of the authors and do not necessarily reflect the views of the publisher.
We thank Barbara Siegfried, Sonja Eisenring, and Nadja Hertel for their support during the laboratory work. This research was funded by the Swiss National Science Foundation (SNSF) under grant no. 188684, as part of the IQ-SASS project (Improved Quantitative Source Assessment of Organic Matter in Soils and Sediments using Molecular Markers and Inverse Modelling). Dario Püntener was supported by a Cotutelle grant by swissuniversities (grant no. NL24/01). We thank the editor Alix Vidal, Jérémy Puissant and an anonymous referee for their feedback on the manuscript.
This research has been supported by the Schweizerischer Nationalfonds zur Förderung der Wissenschaftlichen Forschung (grant no. 188684). Dario Püntener was supported by a Cotutelle grant by swissuniversities (grant no. NL24/01).
This paper was edited by Alix Vidal and reviewed by Jérémy Puissant and one anonymous referee.
Abdalla, K., Schierling, L., Sun, Y., Schuchardt, M. A., Jentsch, A., Deola, T., Wolff, P., Kiese, R., Lehndorff, E., Pausch, J., and Meyer, N.: Temperature sensitivity of soil respiration declines with climate warming in subalpine and alpine grassland soils, Biogeochemistry, 167, 1453–1467, https://doi.org/10.1007/s10533-024-01179-3, 2024. a, b
Abiven, S. and Andreoli, R.: Charcoal does not change the decomposition rate of mixed litters in a mineral cambisol: a controlled conditions study, Biol. Fertil. Soils, 47, 111–114, https://doi.org/10.1007/s00374-010-0489-1, 2011. a, b
Adekanmbi, A. A., Shu, X., Zou, Y., and Sizmur, T.: Legacy effect of constant and diurnally oscillating temperatures on soil respiration and microbial community structure, Eur. J. Soil Sci., 73, https://doi.org/10.1111/ejss.13319, 2022. a
Alster, C. J., van de Laar, A., Goodrich, J. P., Arcus, V. L., Deslippe, J. R., Marshall, A. J., and Schipper, L. A.: Quantifying thermal adaptation of soil microbial respiration, Nat. Commun., 14, 5459, https://doi.org/10.1038/s41467-023-41096-x, 2023. a
Bai, Y., Peng, Y., Zhang, D., Yang, G., Chen, L., Kang, L., Zhou, W., Wei, B., Xie, Y., and Yang, Y.: Heating up the roof of the world: tracing the impacts of in-situ warming on carbon cycle in alpine grasslands on the Tibetan Plateau, Natl. Sci. Rev., 12, nwae371, https://doi.org/10.1093/nsr/nwae371, 2025. a, b
Bastida, F., García, C., Fierer, N., Eldridge, D. J., Bowker, M. A., Abades, S., Alfaro, F. D., Asefaw Berhe, A., Cutler, N. A., Gallardo, A., García-Velázquez, L., Hart, S. C., Hayes, P. E., Hernández, T., Hseu, Z. Y., Jehmlich, N., Kirchmair, M., Lambers, H., Neuhauser, S., Peña-Ramírez, V. M., Pérez, C. A., Reed, S. C., Santos, F., Siebe, C., Sullivan, B. W., Trivedi, P., Vera, A., Williams, M. A., Luis Moreno, J., and Delgado-Baquerizo, M.: Global ecological predictors of the soil priming effect, Nat. Commun., 10, https://doi.org/10.1038/s41467-019-11472-7, 2019. a, b, c, d, e
Bonfanti, N., Clément, J. C., Münkemüller, T., Barré, P., Baudin, F., and Poulenard, J.: Prolonged warming leads to carbon depletion and increases nutrient availability in alpine soils, Appl. Soil Ecol., 213, 106239, https://doi.org/10.1016/j.apsoil.2025.106239, 2025. a, b
Breitenbach, S. F. M. and Bernasconi, S. M.: Carbon and oxygen isotope analysis of small carbonate samples (20 to 100 µg) with a GasBench II preparation device, Rapid Commun. Mass Spectrom., 25, 1910–1914, https://doi.org/10.1002/rcm.5052, 2011. a
Broadbent, A. A. D., Snell, H. S. K., Michas, A., Pritchard, W. J., Newbold, L., Cordero, I., Goodall, T., Schallhart, N., Kaufmann, R., Griffiths, R. I., Schloter, M., Bahn, M., and Bardgett, R. D.: Climate change alters temporal dynamics of alpine soil microbial functioning and biogeochemical cycling via earlier snowmelt, ISME J., 15, 2264–2275, https://doi.org/10.1038/s41396-021-00922-0, 2021. a
Budge, K., Leifeld, J., Hiltbrunner, E., and Fuhrer, J.: Alpine grassland soils contain large proportion of labile carbon but indicate long turnover times, Biogeosciences, 8, 1911–1923, https://doi.org/10.5194/bg-8-1911-2011, 2011. a, b
Chen, J., Luo, Y., García-Palacios, P., Cao, J., Dacal, M., Zhou, X., Li, J., Xia, J., Niu, S., Yang, H., Shelton, S., Guo, W., and van Groenigen, K. J.: Differential responses of carbon-degrading enzyme activities to warming: Implications for soil respiration, Global Change Biol., 24, 4816–4826, https://doi.org/10.1111/gcb.14394, 2018. a, b
Chen, J., Zhang, Y., Kuzyakov, Y., Wang, D., and Olesen, J. E.: Challenges in upscaling laboratory studies to ecosystems in soil microbiology research, Global Change Biol., 29, 569–574, https://doi.org/10.1111/gcb.16537, 2023. a
Chen, Y., Qin, W., Zhang, Q., Wang, X., Feng, J., Han, M., Hou, Y., Zhao, H., Zhang, Z., He, J. S., Torn, M. S., and Zhu, B.: Whole-soil warming leads to substantial soil carbon emission in an alpine grassland, Nat. Commun., 15, https://doi.org/10.1038/s41467-024-48736-w, 2024. a, b, c, d, e, f, g, h
Chersich, S., Rejšek, K., Vranová, V., Bordoni, M., and Meisina, C.: Climate change impacts on the Alpine ecosystem: an overview with focus on the soil, J. Forest Sci., 61, 496–514, https://doi.org/10.17221/47/2015-JFS, 2015. a
Classen, A. T., Sundqvist, M. K., Henning, J. A., Newman, G. S., Moore, J. A., Cregger, M. A., Moorhead, L. C., and Patterson, C. M.: Direct and indirect effects of climate change on soil microbial and soil microbial-plant interactions: What lies ahead?, Ecosphere, 6, https://doi.org/10.1890/ES15-00217.1, 2015. a
Craine, J. M., Fierer, N., and McLauchlan, K. K.: Widespread coupling between the rate and temperature sensitivity of organic matter decay, Nat. Geosci., 3, 854–857, https://doi.org/10.1038/ngeo1009, 2010. a
Creamer, C. A., Jones, D. L., Baldock, J. A., Rui, Y., Murphy, D. V., Hoyle, F. C., and Farrell, M.: Is the fate of glucose-derived carbon more strongly driven by nutrient availability, soil texture, or microbial biomass size?, Soil Biol. Biochem., 103, 201–212, https://doi.org/10.1016/j.soilbio.2016.08.025, 2016. a, b, c, d
Crowther, T. W., Todd-Brown, K. E. O., Rowe, C. W., Wieder, W. R., Carey, J. C., Machmuller, M. B., Snoek, B. L., Fang, S., Zhou, G., Allison, S. D., Blair, J. M., Bridgham, S. D., Burton, A. J., Carrillo, Y., Reich, P. B., Clark, J. S., Classen, A. T., Dijkstra, F. A., Elberling, B., Emmett, B. A., Estiarte, M., Frey, S. D., Guo, J., Harte, J., Jiang, L., Johnson, B. R., Kröel-Dulay, G., Larsen, K. S., Laudon, H., Lavallee, J. M., Luo, Y., Lupascu, M., Ma, L. N., Marhan, S., Michelsen, A., Mohan, J., Niu, S., Pendall, E., Peñuelas, J., Pfeifer-Meister, L., Poll, C., Reinsch, S., Reynolds, L. L., Schmidt, I. K., Sistla, S., Sokol, N. W., Templer, P. H., Treseder, K. K., Welker, J. M., and Bradford, M. A.: Quantifying global soil carbon losses in response to warming, Nature, 540, 104–108, https://doi.org/10.1038/nature20150, 2016. a, b
Cui, J., Ge, T., Nie, M., Kuzyakov, Y., Alharbi, S., Fang, C., and Deng, Z.: Contrasting effects of maize litter and litter-derived biochar on the temperature sensitivity of paddy soil organic matter decomposition, Front. Microbiol., 13, https://doi.org/10.3389/fmicb.2022.1008744, 2022. a
Dang, R., Liu, J., Lichtfouse, E., Zhou, L., Zhou, M., and Xiao, L.: Soil microbial carbon use efficiency and the constraints, Ann. Microbiol., 74, 37, https://doi.org/10.1186/s13213-024-01780-9, 2024. a, b
Davidson, E. A. and Janssens, I. A.: Temperature sensitivity of soil carbon decomposition and feedbacks to climate change, Nature, 440, 165–173, https://doi.org/10.1038/nature04514, 2006. a, b, c, d, e, f, g, h
Dengzeng, Z., Ma, W., Wang, C., Tang, S., and Zhang, D.: Effect of shrub encroachment on alpine grass soil microbial community assembly, Front. Soil Sci., 2, https://doi.org/10.3389/fsoil.2022.829575, 2022. a, b
Dignac, M. F., Bahri, H., Rumpel, C., Rasse, D. P., Bardoux, G., Balesdent, J., Girardin, C., Chenu, C., and Mariotti, A.: Carbon-13 natural abundance as a tool to study the dynamics of lignin monomers in soil: An appraisal at the Closeaux experimental field (France), Geoderma, 128, 3–17, https://doi.org/10.1016/j.geoderma.2004.12.022, 2005. a
Djukic, I., Zehetner, F., Tatzber, M., and Gerzabek, M. H.: Soil organic‐matter stocks and characteristics along an Alpine elevation gradient, J. Plant Nutr. Soil Sci., 173, 30–38, https://doi.org/10.1002/jpln.200900027, 2010. a
Djukic, I., Zehetner, F., Watzinger, A., Horacek, M., and Gerzabek, M. H.: In situ carbon turnover dynamics and the role of soil microorganisms therein: a climate warming study in an Alpine ecosystem, FEMS Microbiol. Ecol., 83, 112–124, https://doi.org/10.1111/j.1574-6941.2012.01449.x, 2013. a
Domeignoz-Horta, L. A., Pold, G., Liu, X. J. A., Frey, S. D., Melillo, J. M., and DeAngelis, K. M.: Microbial diversity drives carbon use efficiency in a model soil, Nat. Commun., 11, https://doi.org/10.1038/s41467-020-17502-z, 2020. a, b
Donhauser, J. and Frey, B.: Alpine soil microbial ecology in a changing world, FEMS Microbiol. Ecol., 94, https://doi.org/10.1093/femsec/fiy099, 2018. a
Donhauser, J., Niklaus, P. A., Rousk, J., Larose, C., and Frey, B.: Temperatures beyond the community optimum promote the dominance of heat-adapted, fast growing and stress resistant bacteria in alpine soils, Soil Biol. Biochem., 148, https://doi.org/10.1016/j.soilbio.2020.107873, 2020. a, b, c, d
Fanin, N., Mooshammer, M., Sauvadet, M., Meng, C., Alvarez, G., Bernard, L., Bertrand, I., Blagodatskaya, E., Bon, L., Fontaine, S., Niu, S., Lashermes, G., Maxwell, T., Weintraub, M. N., Wingate, L., Moorhead, D., and Nottingham, A. T.: Soil enzymes in response to climate warming: Mechanisms and feedbacks, Funct. Ecol., 36, 1378–1395, https://doi.org/10.1111/1365-2435.14027, 2022. a, b
Fierer, N., Craine, J. M., Mclauchlan, K., and Schimel, J. P.: Litter quality and the temperature sensitivity of decomposition, Ecology, 86, 320–326, https://doi.org/10.1890/04-1254, 2005. a
Frey, S. D., Lee, J., Melillo, J. M., and Six, J.: The temperature response of soil microbial efficiency and its feedback to climate, Nat. Clim. Change, 3, 395–398, https://doi.org/10.1038/nclimate1796, 2013. a, b, c
Friedlingstein, P., O'Sullivan, M., Jones, M. W., Andrew, R. M., Hauck, J., Landschützer, P., Le Quéré, C., Li, H., Luijkx, I. T., Olsen, A., Peters, G. P., Peters, W., Pongratz, J., Schwingshackl, C., Sitch, S., Canadell, J. G., Ciais, P., Jackson, R. B., Alin, S. R., Arneth, A., Arora, V., Bates, N. R., Becker, M., Bellouin, N., Berghoff, C. F., Bittig, H. C., Bopp, L., Cadule, P., Campbell, K., Chamberlain, M. A., Chandra, N., Chevallier, F., Chini, L. P., Colligan, T., Decayeux, J., Djeutchouang, L. M., Dou, X., Duran Rojas, C., Enyo, K., Evans, W., Fay, A. R., Feely, R. A., Ford, D. J., Foster, A., Gasser, T., Gehlen, M., Gkritzalis, T., Grassi, G., Gregor, L., Gruber, N., Gürses, Ö., Harris, I., Hefner, M., Heinke, J., Hurtt, G. C., Iida, Y., Ilyina, T., Jacobson, A. R., Jain, A. K., Jarníková, T., Jersild, A., Jiang, F., Jin, Z., Kato, E., Keeling, R. F., Klein Goldewijk, K., Knauer, J., Korsbakken, J. I., Lan, X., Lauvset, S. K., Lefèvre, N., Liu, Z., Liu, J., Ma, L., Maksyutov, S., Marland, G., Mayot, N., McGuire, P. C., Metzl, N., Monacci, N. M., Morgan, E. J., Nakaoka, S.-I., Neill, C., Niwa, Y., Nützel, T., Olivier, L., Ono, T., Palmer, P. I., Pierrot, D., Qin, Z., Resplandy, L., Roobaert, A., Rosan, T. M., Rödenbeck, C., Schwinger, J., Smallman, T. L., Smith, S. M., Sospedra-Alfonso, R., Steinhoff, T., Sun, Q., Sutton, A. J., Séférian, R., Takao, S., Tatebe, H., Tian, H., Tilbrook, B., Torres, O., Tourigny, E., Tsujino, H., Tubiello, F., van der Werf, G., Wanninkhof, R., Wang, X., Yang, D., Yang, X., Yu, Z., Yuan, W., Yue, X., Zaehle, S., Zeng, N., and Zeng, J.: Global Carbon Budget 2024, Earth Syst. Sci. Data, 17, 965–1039, https://doi.org/10.5194/essd-17-965-2025, 2025. a
Frostegård, Å., Tunlid, A., and Bååth, E.: Microbial biomass measured as total lipid phosphate in soils of different organic content, J. Microbiol. Meth., 14, 151–163, https://doi.org/10.1016/0167-7012(91)90018-L, 1991. a
García-Palacios, P., Crowther, T. W., Dacal, M., Hartley, I. P., Reinsch, S., Rinnan, R., Rousk, J., van den Hoogen, J., Ye, J.-S., and Bradford, M. A.: Evidence for large microbial-mediated losses of soil carbon under anthropogenic warming, Nat. Rev. Earth Environ., 2, 507–517, https://doi.org/10.1038/s43017-021-00178-4, 2021. a, b, c
Gavazov, K., Ingrisch, J., Hasibeder, R., Mills, R. T., Buttler, A., Gleixner, G., Pumpanen, J., and Bahn, M.: Winter ecology of a subalpine grassland: Effects of snow removal on soil respiration, microbial structure and function, Sci. Total Environ., 590-591, 316–324, https://doi.org/10.1016/j.scitotenv.2017.03.010, 2017. a
Gehrig‐Fasel, J., Guisan, A., and Zimmermann, N. E.: Tree line shifts in the Swiss Alps: Climate change or land abandonment?, J. Veg. Sci., 18, 571–582, https://doi.org/10.1111/j.1654-1103.2007.tb02571.x, 2007. a, b, c
Gershenson, A., Bader, N. E., and Cheng, W.: Effects of substrate availability on the temperature sensitivity of soil organic matter decomposition, Global Change Biol., 15, 176–183, https://doi.org/10.1111/j.1365-2486.2008.01827.x, 2009. a
Gunina, A., Dippold, M., Glaser, B., and Kuzyakov, Y.: Turnover of microbial groups and cell components in soil: 13C analysis of cellular biomarkers, Biogeosciences, 14, 271–283, https://doi.org/10.5194/bg-14-271-2017, 2017a. a
Gunina, A., Smith, A. R., Godbold, D. L., Jones, D. L., and Kuzyakov, Y.: Response of soil microbial community to afforestation with pure and mixed species, Plant Soil, 412, 357–368, https://doi.org/10.1007/s11104-016-3073-0, 2017b. a, b, c
Hagedorn, F., Mulder, J., and Jandl, R.: Mountain soils under a changing climate and land-use, Biogeochemistry, 97, 1–5, https://doi.org/10.1007/s10533-009-9386-9, 2010. a
Hagedorn, F., Gavazov, K., and Alexander, J. M.: Above- and belowground linkages shape responses of mountain vegetation to climate change, Science, 365, 1119–1123, https://doi.org/10.1126/science.aax4737, 2019. a, b, c, d
Hanson, P. J., Griffiths, N. A., Iversen, C. M., Norby, R. J., Sebestyen, S. D., Phillips, J. R., Chanton, J. P., Kolka, R. K., Malhotra, A., Oleheiser, K. C., Warren, J. M., Shi, X., Yang, X., Mao, J., and Ricciuto, D. M.: Rapid net carbon loss from a whole‐ecosystem warmed peatland, AGU Adv., 1, https://doi.org/10.1029/2020av000163, 2020. a, b
Hiltbrunner, D., Zimmermann, S., and Hagedorn, F.: Afforestation with Norway spruce on a subalpine pasture alters carbon dynamics but only moderately affects soil carbon storage, Biogeochemistry, 115, 251–266, https://doi.org/10.1007/s10533-013-9832-6, 2013. a, b, c
Hock, R., Rasul, G., Adler, C., Cáceres, B., Gruber, S., Hirabayashi, Y., Jackson, M., Kääb, A., Kang, S., Kutuzov, S., Milner, A., Molau, U., Morin, S., Orlove, B., and Steltzer, H.: High Mountain Areas, in: The Ocean and Cryosphere in a Changing Climate, Cambridge University Press, 131–202, https://doi.org/10.1017/9781009157964.004, 2019. a, b
IUSS Working Group WRB: World Reference Base for Soil Resources 2014, update 2015, International soil classification system for naming soils and creating legends for soil maps, Tech. rep., FAO, Rome, ISBN 978-92-5-108369-7, 2015. a
Klein, G., Vitasse, Y., Rixen, C., Marty, C., and Rebetez, M.: Shorter snow cover duration since 970 in the Swiss Alps due to earlier snowmelt more than to later snow onset, Climatic Change, 139, 637–649, https://doi.org/10.1007/s10584-016-1806-y, 2016. a
Kramer, C. and Gleixner, G.: Soil organic matter in soil depth profiles: Distinct carbon preferences of microbial groups during carbon transformation, Soil Biol. Biochem., 40, 425–433, https://doi.org/10.1016/j.soilbio.2007.09.016, 2008. a, b, c, d, e
Kravchenko, I. K., Tikhonova, E. N., Ulanova, R. V., Menko, E. V., and Sukhacheva, M. V.: Effect of temperature on litter decomposition, soil microbial community structure and biomass in a mixed-wood forest in European Russia, Current Sci., 116, 765, https://doi.org/10.18520/cs/v116/i5/765-772, 2019. a
Kuzyakov, Y.: Priming effects: Interactions between living and dead organic matter, Soil Biol. Biochem., 42, 1363–1371, https://doi.org/10.1016/j.soilbio.2010.04.003, 2010. a, b
Kuzyakov, Y., Friedel, J., and Stahr, K.: Review of mechanisms and quantification of priming effects, Soil Biol. Biochem., 32, 1485–1498, https://doi.org/10.1016/S0038-0717(00)00084-5, 2000. a, b, c, d
Laorden-Camacho, L., Grigulis, K., Tello-García, E., Lyonnard, B., Colace, M. P., Gallet, C., Tappeiner, U., Leitinger, G., and Lavorel, S.: Shrub encroachment modifies soil properties through plant resource economics traits, Plant Soil, 514, 2083–2104, https://doi.org/10.1007/s11104-025-07506-3, 2025. a, b
Li, J., Wang, G., Mayes, M. A., Allison, S. D., Frey, S. D., Shi, Z., Hu, X., Luo, Y., and Melillo, J. M.: Reduced carbon use efficiency and increased microbial turnover with soil warming, Global Change Biol., 25, 900–910, https://doi.org/10.1111/gcb.14517, 2019. a
Liu, L., Sayer, E. J., Deng, M., Li, P., Liu, W., Wang, X., Yang, S., Huang, J., Luo, J., Su, Y., Grünzweig, J. M., Jiang, L., Hu, S., and Piao, S.: The grassland carbon cycle: Mechanisms, responses to global changes, and potential contribution to carbon neutrality, Fundament. Res., 3, 209–218, https://doi.org/10.1016/j.fmre.2022.09.028, 2023. a, b
Lu, W., Liu, N., Zhang, Y., Zhou, J., Guo, Y., and Yang, X.: Impact of vegetation community on litter decomposition: Evidence from a reciprocal transplant study with 13C labeled plant litter, Soil Biol. Biochem., 112, 248–257, https://doi.org/10.1016/j.soilbio.2017.05.014, 2017. a, b, c, d
Melillo, J. M., Frey, S. D., DeAngelis, K. M., Werner, W. J., Bernard, M. J., Bowles, F. P., Pold, G., Knorr, M. A., and Grandy, A. S.: Long-term pattern and magnitude of soil carbon feedback to the climate system in a warming world, Science, 358, 101–105, https://doi.org/10.1126/science.aan2874, 2017. a, b, c
Merbold, L., Steinlin, C., and Hagedorn, F.: Winter greenhouse gas fluxes (CO2, CH4 and N2O) from a subalpine grassland, Biogeosciences, 10, 3185–3203, https://doi.org/10.5194/bg-10-3185-2013, 2013. a
Moinet, G. Y., Hunt, J. E., Kirschbaum, M. U., Morcom, C. P., Midwood, A. J., and Millard, P.: The temperature sensitivity of soil organic matter decomposition is constrained by microbial access to substrates, Soil Biol. Biochem., 116, 333–339, https://doi.org/10.1016/j.soilbio.2017.10.031, 2018. a, b
Nottingham, A. T., Whitaker, J., Ostle, N. J., Bardgett, R. D., McNamara, N. P., Fierer, N., Salinas, N., Ccahuana, A. J., Turner, B. L., and Meir, P.: Microbial responses to warming enhance soil carbon loss following translocation across a tropical forest elevation gradient, Ecol. Lett., 22, 1889–1899, https://doi.org/10.1111/ele.13379, 2019. a, b
Nottingham, A. T., Scott, J. J., Saltonstall, K., Broders, K., Montero-Sanchez, M., Püspök, J., Bååth, E., and Meir, P.: Microbial diversity declines in warmed tropical soil and respiration rise exceed predictions as communities adapt, Nat. Microbiol., 7, 1650–1660, https://doi.org/10.1038/s41564-022-01200-1, 2022. a, b
Nyberg, M. and Hovenden, M. J.: Warming increases soil respiration in a carbon-rich soil without changing microbial respiratory potential, Biogeosciences, 17, 4405–4420, https://doi.org/10.5194/bg-17-4405-2020, 2020. a, b
Ofiti, N. O., Zosso, C. U., Soong, J. L., Solly, E. F., Torn, M. S., Wiesenberg, G. L., and Schmidt, M. W.: Warming promotes loss of subsoil carbon through accelerated degradation of plant-derived organic matter, Soil Biol. Biochem., 156, 108185, https://doi.org/10.1016/j.soilbio.2021.108185, 2021. a
Ofiti, N. O., Solly, E. F., Hanson, P. J., Malhotra, A., Wiesenberg, G. L., and Schmidt, M. W.: Warming and elevated CO2 promote rapid incorporation and degradation of plant-derived organic matter in an ombrotrophic peatland, Global Change Biol., 28, 883–898, https://doi.org/10.1111/gcb.15955, 2022. a, b
Ofiti, N. O. E., Schmidt, M. W. I., Abiven, S., Hanson, P. J., Iversen, C. M., Wilson, R. M., Kostka, J. E., Wiesenberg, G. L. B., and Malhotra, A.: Climate warming and elevated CO2 alter peatland soil carbon sources and stability, Nat. Commun., 14, 7533, https://doi.org/10.1038/s41467-023-43410-z, 2023. a
Ortiz, C., Vázquez, E., Rubio, A., Benito, M., Schindlbacher, A., Jandl, R., Butterbach-Bahl, K., and Díaz-Pinés, E.: Soil organic matter dynamics after afforestation of mountain grasslands in both a Mediterranean and a temperate climate, Biogeochemistry, 131, 267–280, https://doi.org/10.1007/s10533-016-0278-5, 2016. a, b
Peplau, T., Poeplau, C., Gregorich, E., and Schroeder, J.: Deforestation for agriculture leads to soil warming and enhanced litter decomposition in subarctic soils, Biogeosciences, 20, 1063–1074, https://doi.org/10.5194/bg-20-1063-2023, 2023. a, b
Poeplau, C., Sigurdsson, P., and Sigurdsson, B. D.: Depletion of soil carbon and aggregation after strong warming of a subarctic Andosol under forest and grassland cover, SOIL, 6, 115–129, https://doi.org/10.5194/soil-6-115-2020, 2020. a, b, c
Pold, G., Domeignoz-Horta, L. A., Morrison, E. W., Frey, S. D., Sistla, S. A., and Deangelis, K. M.: Carbon use efficiency and its temperature sensitivity covary in soil bacteria, mBio, 11, https://doi.org/10.1128/mBio.02293-19, 2020. a, b
Püntener, D., Speckert, T. C., Brügger, Y.-A., and Wiesenberg, G. L. B.: Availability of labile carbon controls the temperature-dependent response of soil organic matter decomposition in alpine soils, SOIL, 11, 991–1006, https://doi.org/10.5194/soil-11-991-2025, 2025. a, b, c, d, e, f
Püntener, D., Zürcher, P., Speckert, T. C., Thomas, C. L., and Wiesenberg, G. L. B.: Dataset to Manuscript: Vulnerability of carbon in subalpine soils in the face of warmer temperatures, Zenodo [data set], https://doi.org/10.5281/zenodo.20094476, 2026. a
Qi, Q., Haowei, Y., Zhang, Z., Van Nostrand, J. D., Wu, L., Guo, X., Feng, J., Wang, M., Yang, S., Zhao, J., Gao, Q., Zhang, Q., Zhao, M., Xie, C., Ma, Z., He, J.-S., Chu, H., Huang, Y., Zhou, J., and Yang, Y.: Microbial functional responses explain alpine soil carbon fluxes under future climate scenarios, mBio, 12, 1–13, https://doi.org/10.1128/mBio.00761-20, 2021. a
R Core Team: R: A language and environment for statistical computing, https://www.R-project.org/ (last access: 9 May 2026), 2025. a
Rindt, O., Rosinger, C., Bonkowski, M., Rixen, C., Brüggemann, N., Urich, T., and Fiore-Donno, A. M.: Biogeochemical dynamics during snowmelt and in summer in the Alps, Biogeochemistry, 162, 257–266, https://doi.org/10.1007/s10533-022-01005-8, 2023. a
Rogora, M., Frate, L., Carranza, M. L., Freppaz, M., Stanisci, A., Bertani, I., Bottarin, R., Brambilla, A., Canullo, R., Carbognani, M., Cerrato, C., Chelli, S., Cremonese, E., Cutini, M., Di Musciano, M., Erschbamer, B., Godone, D., Iocchi, M., Isabellon, M., Magnani, A., Mazzola, L., Morra di Cella, U., Pauli, H., Petey, M., Petriccione, B., Porro, F., Psenner, R., Rossetti, G., Scotti, A., Sommaruga, R., Tappeiner, U., Theurillat, J. P., Tomaselli, M., Viglietti, D., Viterbi, R., Vittoz, P., Winkler, M., and Matteucci, G.: Assessment of climate change effects on mountain ecosystems through a cross-site analysis in the Alps and Apennines, Sci. Total Environ., 624, 1429–1442, https://doi.org/10.1016/j.scitotenv.2017.12.155, 2018. a, b
Schiedung, M., Don, A., Beare, M. H., and Abiven, S.: Soil carbon losses due to priming moderated by adaptation and legacy effects, Nat. Geosci., 16, 909–914, https://doi.org/10.1038/s41561-023-01275-3, 2023. a, b
Schindlbacher, A., Rodler, A., Kuffner, M., Kitzler, B., Sessitsch, A., and Zechmeister-Boltenstern, S.: Experimental warming effects on the microbial community of a temperate mountain forest soil, Soil Biol. Biochem., 43, 1417–1425, https://doi.org/10.1016/j.soilbio.2011.03.005, 2011. a, b, c, d
Schmidt, M. W. I., Torn, M. S., Abiven, S., Dittmar, T., Guggenberger, G., Janssens, I. A., Kleber, M., Kögel-Knabner, I., Lehmann, J., Manning, D. A. C., Nannipieri, P., Rasse, D. P., Weiner, S., and Trumbore, S. E.: Persistence of soil organic matter as an ecosystem property, Nature, 478, 49–56, https://doi.org/10.1038/nature10386, 2011. a
Schwörer, C., Colombaroli, D., Kaltenrieder, P., Rey, F., and Tinner, W.: Early human impact (5000–3000 BC) affects mountain forest dynamics in the Alps, J. Ecol., 103, 281–295, https://doi.org/10.1111/1365-2745.12354, 2015. a
Slater, C., Preston, T., and Weaver, L. T.: Stable isotopes and the international system of units, Rapid Commun. Mass Spectrom., 15, 1270–1273, https://doi.org/10.1002/rcm.328, 2001. a
Solly, E. F., Schöning, I., Boch, S., Kandeler, E., Marhan, S., Michalzik, B., Müller, J., Zscheischler, J., Trumbore, S. E., and Schrumpf, M.: Factors controlling decomposition rates of fine root litter in temperate forests and grasslands, Plant Soil, 382, 203–218, https://doi.org/10.1007/s11104-014-2151-4, 2014. a, b, c
Soong, J. L., Castanha, C., Hicks Pries, C. E., Ofiti, N., Porras, R. C., Riley, W. J., Schmidt, M. W., and Torn, M. S.: Five years of whole-soil warming led to loss of subsoil carbon stocks and increased CO2 efflux, Sci. Adv., 7, 1–8, https://doi.org/10.1126/sciadv.abd1343, 2021. a, b, c
Speckert, T. C., Suremann, J., Gavazov, K., Santos, M. J., Hagedorn, F., and Wiesenberg, G. L. B.: Soil organic carbon stocks did not change after 130 years of afforestation on a former Swiss Alpine pasture, SOIL, 9, 609–621, https://doi.org/10.5194/soil-9-609-2023, 2023. a, b, c, d, e
Streit, K., Hagedorn, F., Hiltbrunner, D., Portmann, M., Saurer, M., Buchmann, N., Wild, B., Richter, A., Wipf, S., and Siegwolf, R. T.: Soil warming alters microbial substrate use in alpine soils, Global Change Biol., 20, 1327–1338, https://doi.org/10.1111/gcb.12396, 2014. a, b
Tao, X., Yang, Z., Feng, J., Jian, S., Yang, Y., Bates, C. T., Wang, G., Guo, X., Ning, D., Kempher, M. L., Liu, X. J. A., Ouyang, Y., Han, S., Wu, L., Zeng, Y., Kuang, J., Zhang, Y., Zhou, X., Shi, Z., Qin, W., Wang, J., Firestone, M. K., Tiedje, J. M., and Zhou, J.: Experimental warming accelerates positive soil priming in a temperate grassland ecosystem, Nat. Commun., 15, https://doi.org/10.1038/s41467-024-45277-0, 2024. a, b, c, d, e
Verbrigghe, N., Leblans, N. I., Sigurdsson, B. D., Vicca, S., Fang, C., Fuchslueger, L., Soong, J. L., Weedon, J. T., Poeplau, C., Ariza-Carricondo, C., Bahn, M., Guenet, B., Gundersen, P., Gunnarsdóttir, G. E., Kätterer, T., Liu, Z., Maljanen, M., Marañón-Jiménez, S., Meeran, K., Oddsdóttir, E. S., Ostonen, I., Peñuelas, J., Richter, A., Sardans, J., Sigurðsson, P., Torn, M. S., Van Bodegom, P. M., Verbruggen, E., Walker, T. W., Wallander, H., and Janssens, I. A.: Soil carbon loss in warmed subarctic grasslands is rapid and restricted to topsoil, Biogeosciences, 19, 3381–3393, https://doi.org/10.5194/bg-19-3381-2022, 2022. a
Waldrop, M. P. and Firestone, M. K.: Seasonal dynamics of microbial community composition and function in oak canopy and open grassland soils, Microb. Ecol., 52, 470–479, https://doi.org/10.1007/s00248-006-9100-6, 2006. a
Walker, T. W., Gavazov, K., Guillaume, T., Lambert, T., Mariotte, P., Routh, D., Signarbieux, C., Block, S., Münkemüller, T., Nomoto, H., Crowther, T. W., Richter, A., Buttler, A., and Alexander, J. M.: Lowland plant arrival in alpine ecosystems facilitates a decrease in soil carbon content under experimental climate warming, eLife, 11, https://doi.org/10.7554/eLife.78555, 2022. a, b
Wang, M., Guo, X., Zhang, S., Xiao, L., Mishra, U., Yang, Y., Zhu, B., Wang, G., Mao, X., Qian, T., Jiang, T., Shi, Z., and Luo, Z.: Global soil profiles indicate depth-dependent soil carbon losses under a warmer climate, Nat. Commun., 13, https://doi.org/10.1038/s41467-022-33278-w, 2022. a
Wang, Q., Liu, S., and Tian, P.: Carbon quality and soil microbial property control the latitudinal pattern in temperature sensitivity of soil microbial respiration across Chinese forest ecosystems, Global Change Biol., 24, 2841–2849, https://doi.org/10.1111/gcb.14105, 2018. a
Wang, Y., Xiao, J., Ma, Y., Ding, J., Chen, X., Ding, Z., and Luo, Y.: Persistent and enhanced carbon sequestration capacity of alpine grasslands on Earth’s Third Pole, Sci. Adv., 9, 1–15, https://doi.org/10.1126/sciadv.ade6875, 2023. a, b, c
Wiesenberg, G. L. B. and Gocke, M. I.: Analysis of Lipids and Polycyclic Aromatic Hydrocarbons as Indicators of Past and Present (Micro)Biological Activity, in: Hydrocarbon and Lipid Microbiology Protocols, 1st Edn., edited by: McGenity, T. J., Timmis, K. N., and Nogales, B., Springer, Berlin, Heidelberg, 61–91, https://doi.org/10.1007/8623_2015_157, 2017. a
Wild, B., Gentsch, N., Capek, P., Diáková, K., Alves, R. J., Bárta, J., Gittel, A., Hugelius, G., Knoltsch, A., Kuhry, P., Lashchinskiy, N., Mikutta, R., Palmtag, J., Schleper, C., Schnecker, J., Shibistova, O., Takriti, M., Torsvik, V. L., Urich, T., Watzka, M., Šantrūcková, H., Guggenberger, G., and Richter, A.: Plant-derived compounds stimulate the decomposition of organic matter in arctic permafrost soils, Sci. Rep., 6, https://doi.org/10.1038/srep25607, 2016. a, b, c
Willers, C., Jansen van Rensburg, P., and Claassens, S.: Phospholipid fatty acid profiling of microbial communities-a review of interpretations and recent applications, J. Appl. Microbiol., 119, 1207–1218, https://doi.org/10.1111/jam.12902, 2015. a, b, c
Wollum, A. G. and Gomez, J. E.: A conductivity method for measuring microbially evolved carbon dioxide, Ecology, 51, 155–156, https://doi.org/10.2307/1933610, 1970. a
Xu, H., Huang, L., Chen, J., Zhou, H., Wan, Y., Qu, Q., Wang, M., and Xue, S.: Changes in soil microbial activity and their linkages with soil carbon under global warming, Catena, 232, https://doi.org/10.1016/j.catena.2023.107419, 2023. a
Ye, C., Wang, Y., Yan, X., and Guo, H.: Predominant role of air warming in regulating litter decomposition in a Tibetan alpine meadow: A multi-factor global change experiment, Soil Biol. Biochem., 167, https://doi.org/10.1016/j.soilbio.2022.108588, 2022. a
Yuan, M. M., Guo, X., Wu, L., Zhang, Y., Xiao, N., Ning, D., Shi, Z., Zhou, X., Wu, L., Yang, Y., Tiedje, J. M., and Zhou, J.: Climate warming enhances microbial network complexity and stability, Nat. Clim. Change, 11, 343–348, https://doi.org/10.1038/s41558-021-00989-9, 2021. a
Zehnder, T., Lüscher, A., Ritzmann, C., Pauler, C. M., Berard, J., Kreuzer, M., and Schneider, M. K.: Dominant shrub species are a strong predictor of plant species diversity along subalpine pasture-shrub transects, Alp. Bot., 130, 141–156, https://doi.org/10.1007/s00035-020-00241-8, 2020. a, b
Zhang, H., Ding, W., Luo, J., Bolan, N., Yu, H., and Zhu, J.: Temporal responses of microorganisms and native organic carbon mineralization to 13C-glucose addition in a sandy loam soil with long-term fertilization, Eur. J. Soil Biol., 74, 16–22, https://doi.org/10.1016/j.ejsobi.2016.02.007, 2016. a, b
Zheng, T., Miltner, A., Liang, C., Nowak, K. M., and Kästner, M.: Turnover of gram-negative bacterial biomass-derived carbon through the microbial food web of an agricultural soil, Soil Biol. Biochem., 152, https://doi.org/10.1016/j.soilbio.2020.108070, 2021. a, b
Zhu, X., Guo, Z., Wang, N., Liu, J., Zuo, Y., Li, K., Song, C., Song, Y., Gong, C., Xu, X., Yuan, F., and Zhang, L.: Environmental stress stimulates microbial activities as indicated by cyclopropane fatty acid enhancement, Sci. Total Environ., 873, https://doi.org/10.1016/j.scitotenv.2023.162338, 2023. a, b, c
Zosso, C. U. and Wiesenberg, G. L.: Methylation procedures affect PLFA results more than selected extraction parameters, J. Microbiol. Meth., 182, 106164, https://doi.org/10.1016/j.mimet.2021.106164, 2021. a, b, c, d
Zosso, C. U., Ofiti, N. O. E., Soong, J. L., Solly, E. F., Torn, M. S., Huguet, A., Wiesenberg, G. L. B., and Schmidt, M. W. I.: Whole-soil warming decreases abundance and modifies the community structure of microorganisms in the subsoil but not in surface soil, SOIL, 7, 477–494, https://doi.org/10.5194/soil-7-477-2021, 2021. a, b
Zosso, C. U., Ofiti, N. O., Torn, M. S., Wiesenberg, G. L., and Schmidt, M. W.: Rapid loss of complex polymers and pyrogenic carbon in subsoils under whole-soil warming, Nat. Geosci., 16, 344–348, https://doi.org/10.1038/s41561-023-01142-1, 2023. a, b